In the News
What happened in healthcare recently—and what we think about it.
- CMS raises expectations for hospital nutrition services. The Centers for Medicare & Medicaid Services (CMS) this week outlined stricter expectations for hospital (including critical access hospital) nutrition services, with surveyors directed to take a closer look at how facilities assess, plan, and deliver nutritional services. In a memo published Monday, CMS said that nutrition services should be based on clinical needs, with hospitals expected to document patient-specific nutrition assessments and ensure that care plans match each patient’s condition. The memo calls for stronger integration between dietitians and the broader care team, reinforcing that nutrition is part of treatment, not a standalone service. Federal inspectors working on behalf of Medicare will check whether meals support patient health and follow established nutrition guidelines, rather than being based on patient preference or convenience. Federal dietary guidelines, updated in January, urge limiting “highly processed foods” while prioritizing meat and whole-fat dairy products.
- The Gist: The guidance does not create new regulations, but it signals a potentially more rigorous enforcement posture. It’s hard to argue with the goal of serving healthier foods to hospitalized patients. Health systems may want to review the updated dietary guidelines, consider how dietitians are involved in care planning, and identify whether menus align with clinical needs. The guidance also suggests a more consistent approach to balancing patient preference with medical appropriateness, an area that has often been handled informally. Over time, this could lead to more standardized practices across hospitals, particularly as survey activity begins to reflect these expectations.
- FDA approves Lilly weight loss pill, expanding GLP-1 market. A new daily weight loss pill from Eli Lilly won Food and Drug Administration approval on Wednesday, marking an expansion of the fast-growing GLP-1 drug class. The drug, sold as Foundayo, is the second oral GLP-1 treatment cleared for obesity and offers an alternative to widely used injectable medications like Wegovy and Zepbound. Unlike earlier options, the pill can be taken at any time of day without food restrictions, a convenience that could broaden patient uptake. In clinical trials, patients lost roughly 11% of body weight on higher doses, somewhat below leading injectable therapies but still clinically meaningful. The FDA reviewed the drug under an expedited program, underscoring federal interest in accelerating treatments for obesity and related conditions.
- The Gist: Oral medications typically cost less to manufacture and don’t have the same storage and shipping requirements as injectable drugs. They are also more attractive to patients who suffer from “needlephobia.” Oral GLP-1s likely will lower the barrier to entry, which could drive higher utilization. That has downstream implications for demand management, care coordination, and monitoring of side effects, particularly as GLP-1 therapy moves earlier into treatment pathways. At the same time, questions around cost, coverage, and long-term adherence remain unresolved. Easier access to GLP-1s could reshape service line demand over time; they may accelerate declines in bariatric surgery volumes and could affect referral patterns across endocrinology, cardiology, and primary care. Increased uptake could drive more demand for longitudinal management of obesity and related conditions, including diabetes, heart disease, and sleep apnea. Hospitals may need to reassess where weight management “lives” within the organization, whether as part of a metabolic service line, a virtual care model, or embedded in primary care.
- HHS, CMS name advisory committee to reshape healthcare. A new federal advisory group announced last week is intended to inform recommendations on how to modernize the U.S. healthcare system and improve care. The Healthcare Advisory Committee, announced by the Department of Health and Human Services and CMS, will offer nonbinding recommendations on potential changes to how care is financed and delivered across Medicare, Medicaid, the Children’s Health Insurance Program, and the individual market. Members include a mix of health system executives, policy experts, and private-sector leaders. The committee’s agenda spans policies to prevent and manage chronic disease; reducing administrative burden; expanding the use of data to support care, speed claims processing and improve quality measurement; enhancing care for vulnerable populations; and strengthening Medicare Advantage. It is expected to convene its first meeting later this year.
- The Gist: The new committee is advisory in nature, but its mandate—to shape policy around cost, quality and administrative burden—bears watching. The inclusion of health system C-suite leaders may signal a push toward financial discipline, particularly in Medicare Advantage and risk adjustment. At the same time, the committee’s stated goals around real-time data and claims processing could increase focus on operational efficiency and interoperability. One wildcard: motivational speaker Tony Robbins is among the members, an unconventional pick for a federal advisory body. If nothing else, his inclusion may hint at broader ambitions: as Robbins reminds his fans often, “your past does not equal your future.” Whether that translates into actionable policy is a question hospitals will watch closely.
Plus—what we’ve been reading.
- Gray hairs from the explosion of ‘gray-market’ drugs. Published last month in The Atlantic, this article describes the fast-growing, loosely regulated market for compounded peptide drugs that has slipped beyond traditional oversight. Originally intended for niche cases like drug shortages or patients who cannot tolerate standard formulations, compounding pharmacies and telehealth companies have expanded into mass-market offerings, particularly for weight loss, sexual health, and cosmetic uses. Demand for GLP-1 drugs has accelerated this trend, with cheaper compounded versions attracting patients priced out of branded therapies. But many of these products operate in a gray zone: some are lightly modified versions of approved drugs, while others are experimental compounds with little clinical evidence and uncertain contents. Regulators have struggled to keep pace, resulting in what amounts to “whack-a-mole” enforcement in which questionable products remain widely available.
- The Gist: This is foremost a demand signal: patients are willing to bypass traditional care pathways when they perceive that access, cost, or convenience fall short. The growth of the peptide industry also exposes a credibility gap: patients sometimes trust telehealth startups and online pharmacies over clinicians. Regulation alone is powerless to bridge that gap; the market has spoken. At the same time, the clinical and legal risks are substantial, given that many unapproved peptides are high-risk substances. Health systems will need to decide whether and how to engage—by offering safer alternatives, clear guidance, and easier access. Ignoring the trend is the least viable option; patients are voting with their feet (and with their wallets), whether we like it or not.
Graphic of the Week
A key insight illustrated in infographic form.
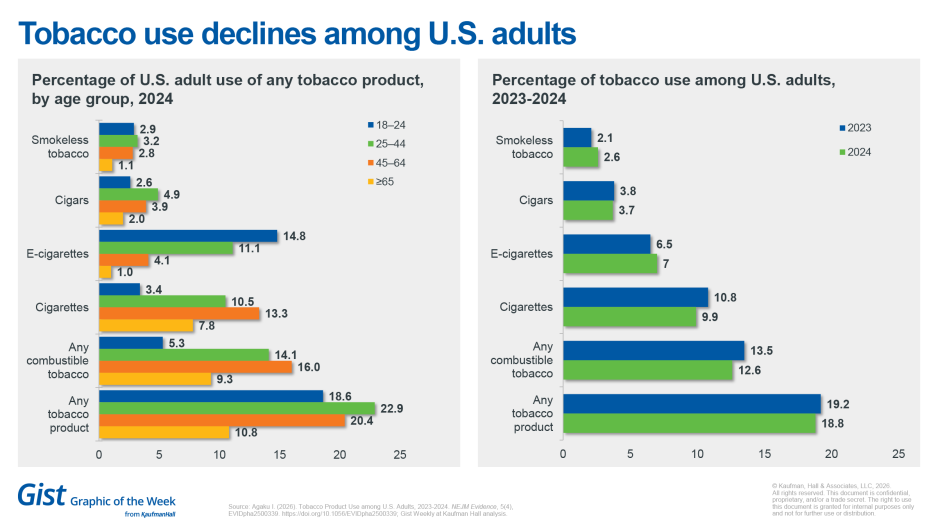
Tobacco use falls into single digits
New research published in NEJM Evidence finds tobacco use continued to decline among adults in the United States from 2023 to 2024. The decline in cigarette smoking from 10.8% in 2023 to 9.9% in 2024 marked the first time that the prevalence of cigarette smoking among U.S. adults reached single digits. This aligns with progress toward the Healthy People 2030 goal of 6.1% and indicates that the goal may be met or exceeded by 2030. However, the rise in smokeless tobacco and e-cigarette use and lack of change in cigar use suggest that tobacco control approaches are not universally successful. In this study, drawn from 2023 and 2024 National Health Interview Survey data, the author observes that routine public reporting of adult smoking prevalence has been interrupted, creating gaps in the continuity of federal tobacco surveillance.
This Week at Kaufman Hall
What our experts are saying about key issues in healthcare.
In a healthcare market where reimbursement pressures and policy uncertainty outpace growth, non-acute partnerships are an emerging area of opportunity for providers.
A new article explores how health systems are using non-acute partnerships to move faster, respond to market pressure, and create value beyond traditional hospital expansion.
On Our Podcast
The Gist Healthcare Podcast—all the headlines in healthcare policy, business and more, in 10 minutes or less every other weekday morning.
Earlier this week, host J. Carlisle Larsen spoke with Phil Englert of Health-ISAC about the cyberattack on medical device giant Stryker, an unusual and disruptive incident that didn’t follow the typical ransomware playbook. Across the two-part conversation, they examined what made the attack different, how healthcare organizations responded in real time, and what it reveals about device security and industry coordination.
Coming up next Monday, health systems are adding more advanced practice providers (APPs), but many aren’t seeing the payoff. In an encore presentation, Kaufman Hall’s Bonnie Proulx joins JC to unpack why physician-APP models often fall short, and what leaders can do to make team-based care work.
To stay up to date, be sure to tune in every Monday, Wednesday and Friday morning. Subscribe on Apple, Spotify, Google or wherever podcasts are available.
Thanks for reading! We’ll see you next Friday with a new edition. In the meantime, check out our Gist Weekly archive for past editions. We also have all of our recent Graphics of the Week available.
Best regards,
The Gist Weekly team at Kaufman Hall